
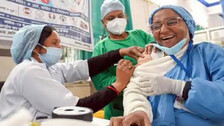
An analysis reveals that Odisha has very fewer vaccination sites to inoculate its eligible population. Chhattisgarh and Assam have opened 4,300 and 3,489 vaccination sites, respectively, against a mere 2,194 in Odisha
Citing the reasons for booster dose in the US, he said that the Delta variant is causing infections in European countries and that is also a reason for booster dose there. However, India has passed that phase now. "Healthcare workers were the first to be vaccinated. But no case has yet come after around 9 to 10 months which can advocate the need for the booster doses in India", opined Dr Seth.
Meanwhile, a total of 110 countries have recognised India's vaccines, Covishield and Covaxin, and agreed to the mutual recognition of Covid-19 vaccination certificates with India.

He inaugurated the first Global Innovation Summit of the pharmaceuticals sector on Thursday. Union Minister Dr. Mansukh Mandaviya was also present on the occasion.

Noting that India has a large pool of scientists and technologists with a potential to take the industry to greater heights, Modi said this strength needs to be harnessed to discover and make in India .

The Union Health Minister expressed confidence that the country will have vaccinated every Indian by the end of the month-long 'Har Ghar Dastak' campaign, according to the statement.

The phase 3 trial Efficacy and Safety Study involving 25,800 volunteers across 25 sites in India is India's largest ever clinical trial conducted for a Covid-19 vaccine.

Moderna's is of the new class of vaccines that use what is called messenger RNA (mRNA) that cause the body to create some proteins similar to that in the Covid-19 virus and trigger the body's immune system to make antibodies that will fight the coronavirus if it invades the body.

Covaxin uses the same Vero Cell manufacturing platform as other childhood vaccines, including the inactivated polio vaccine.

"The vaccine is one-third the adult dose and the vaccine would be given in two doses, three weeks apart. "

For the study, published in The Lancet journal, the team reviewed data from 7,28,321 individuals aged 12 or above who had received the third dose of the BNT162b2 vaccine.

Hyderabad based Bharat Biotech is expected to submit the data by this weekend.

The top court told the petitioner's counsel that it finds no error with the high court decision in the matter, and it will not entertain the petition.
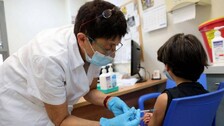
If authorised, Pfizer-BioNTech vaccine would be the first Covid-19 vaccine for younger children.

Last week, the US drugmaker and its German partner BioNTech, in a study, showed that their mRNA Covid vaccine is safe, well tolerated and produces robust neutralising antibody responses in children aged 5 to 11 years.

Copyright © 2024 - Odisha Television Limited All Rights Reserved.