

Hyderabad based Bharat Biotech is expected to submit the data by this weekend.

The top court told the petitioner's counsel that it finds no error with the high court decision in the matter, and it will not entertain the petition.
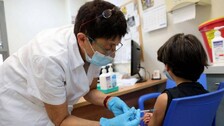
If authorised, Pfizer-BioNTech vaccine would be the first Covid-19 vaccine for younger children.

Last week, the US drugmaker and its German partner BioNTech, in a study, showed that their mRNA Covid vaccine is safe, well tolerated and produces robust neutralising antibody responses in children aged 5 to 11 years.

On Monday, India said it will resume export of surplus COVID-19 vaccines in the fourth quarter of 2021 under the ''Vaccine Maitri'' programme and to meet its commitment to the COVAX global pool.

Stating that there is no shortage of vaccines in the country, the source said that the Centre is capable of supplying as many vaccine doses the states need, in real-time without any delay.

Earlier, Union Minister for Civil Aviation Jyotiraditya Scindia had launched the first of its kind ‘Medicines from the Sky' project at Vikarabad in Telangana under which drugs and vaccines were to be delivered using drones on September 11.

The countrywide vaccination drive was rolled out on January 16 with healthcare workers getting inoculated in the first phase. Vaccination of frontline workers started on February 2.

Moderna is developing vaccines against viral diseases including vaccines against acute respiratory infections, vaccines against persistent infections, as well as vaccines against threats to global public health.

They aim to avoid the use of irregular or suspect products," Anvisa said. The lack of information about the environment at the production bases, combined with the need for vaccine shots to be made in strictly aseptic settings, persuaded officials to take the measure, Anvisa said.

Bhushan also discussed the details of the states and UT's balance stock of 0.5 ml syringes, vaccination coverage among special groups (Transgender Persons, Persons with Disabilities, PWI and Prisoners), Covid vaccination coverage among women particularly pregnant and lactating.

The government in May had further extended the gap between the two doses of Covishield from 6-8 weeks to 12-16 weeks and said the decision to increase the gap was based on scientific evidence.

Although laboratory evidence suggests that antibody responses following Covid-19 vaccination provide better neutralisation of some circulating variants than does natural infection, few real-world epidemiologic studies exist to support the benefit of vaccination for previously infected persons.

President Xi, in a written speech delivered at the first International Forum on COVID-19 Vaccine Cooperation, said that China will provide a total of two billion doses of COVID-19 vaccines to the world within the year to ensure accessibility and affordability of vaccines.

Moderna has also filed to the US Food and Drug Administration for a final approval of its vaccine on June 1, and expects to complete its submission in August.

Copyright © 2024 - Odisha Television Limited All Rights Reserved.